In chemical nomenclature, the IUPAC nomenclature of organic chemistry is a systematic. To avoid long and tedious names in normal communication, the official IUPAC naming recommendations are not always followed in practice, except.
Pretty much every organic chemistry course will start off with three topics - structure, bonding and nomenclature. And, let's face it - it isn't the most interesting topic! It's like learning another language - there's new words, prefixes, suffixes and even grammar. Unfortunately, it's necessary medicine.
If organic chemists don't speak the same language, things are going to get confusing real quick. More importantly to you, as a student, it's definitely going to be on the exam.
Both explicitly, eg. 'Name this compound.' , and as an important part of many other questions, eg. 'What is the product formed when m-chlorobenzoyl chloride reacts with N-ethylisopropylamine?' So it pays to become a Nomenclature Ninja!
You know the English spelling rule - 'i' before 'e', except after 'c'. Except, for the! (seriously, this rule is very flaky.) Well, the language of organic chemistry is also guilty! You know how we said earlier that prefixes like 'di', 'tri' and 'tetra' don't count when alphabetizing? Well, neither do the prefixes in front of common substituents, like tert - and sec.
'iso' isn't counted as a prefix! So, in terms of alphabetical order, 'isobutyl' starts with an 'i', whereas ' sec-butyl' and ' tert-butyl' both start with the letter 'b'.
Don't ask me why - I have no idea! But, we're stuck with it! Here's a final example, this time including substituents with common names.
OFFERED PRICE: Rs. 2,120. Nomenclature of Organic Compounds Table of Content. IUPAC System International union of pure and applied chemistry The most important feature of this system is that any given molecular structure has only one IUPAC name which denotes only one molecular structure. Salient Features of IUPAC System 1. A given compound can be assigned only one name.
A given name can clearly direct in writing of one and only one molecular structure. The system can be applied in naming complex organic compounds. The system can be applied in naming multifunctional organic compounds. This is simple, systematic and scientific method of nomenclature of organic compounds.
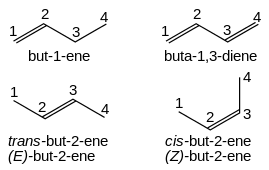
Rule For Naming Prefix (alphabetically) root word (alk) primary suffix (ene, yne) secondary suffix (main functional group) So IUPAC name of any organic compounds essentially consists of two or three parts. (i) Root word (ii) Suffix (iii) Prefix (i) Root Words The basic unit is a series of root words which indicate linear or continuous chains of carbon atoms. Chains containing one to four carbon atoms are known by special root words while chains from C 5 onwards are known by Greek number roots. Chain Length Root word Chain Length Root word C 1 Meth- C 11 Undec- C 2 Eth- C 12 Dodec- C 2 Prop- C 13 Tridec- C 4 But- C 14 Tetradec- C 5 Pent- C 15 Pentadec- C 6 Hex- C 16 Hexadec- C 7 Hept- C 20 Eicos- C 8 Oct- C 30 Triacont- C 9 Non- C 40 Tetracont- C 10 Dec- C 50 Pentacont- In general, the root word for any carbon chain in alk. (ii) Primary Suffix Primary suffix are added to the root words to show saturation or unsaturation in a carbon chain. Nature of carbon chain Primary suffix Generic name Saturated (C – C) -ane Alkane Unsaturated (C = C) with one double bond -ene Alkene Unsaturated (C º C) with one triple bond -yne Alkyne Unsaturated with two C = C bonds -diene Akladiene Unsaturated with two C º C bonds -diyne Alkadiyne Unsaturated with three C = C bonds -triene Alkatriene (iii) Secondary Suffixes Suffixes added after the primary suffix to indicate the presence of a particular functional group in the carbon chain are known as secondary suffixes. Functional Group Secondary suffix Alcohol (-OH) -ol Aldehyde (-CHO) -al Ketone (CO) -one- Carboxylic acid (-COOH) -oic acid Sulphonic (-SO 3H) -sulphonic acid Amine (-NH 2) -amine Thioalcohol (-SH) -thiol Cyanide (-CN) -nitrile Ester (-COOR) -oate Amide (-CONH 2) -amide Acid halide (-COX) -oyl halide Note: The terminal ‘e’ of the primary suffix is removed when initial letter of secondary suffix is vowel.
